Previously 21 months since Covid-19 grew to become a public well being disaster, adoption of digital well being options has accelerated in an effort to mitigate danger. Though as soon as thought to be a helpful companion to in-patient visits, digital care as a default place to begin for healthcare supply is changing into more and more frequent, due to CMS reimbursement. And that’s changing into the norm not solely for a lot of main and a few pressing care supply, but additionally for specialty areas resembling behavioral well being. Nationwide retail chains resembling CVS Well being and Walgreens have adopted platforms to assist Digital First Care (Virtual1Care). Amazon Care embraces Virtual1Care – it’s made the service out there to its workers throughout the nation and different firms are starting to make use of it as nicely, significantly Hilton.
Virtual1Care is a part of a wider pattern we’re seeing within the restructuring of healthcare. As a way to profit from this pattern, pharma and medtech firms should be sure that their digital well being instruments, wrapped round a drug or system, adjust to evolving rules. In an interview, BrightInsight CEO and Founder, Dr. Kal Patel mentioned how Virtual1Care is shaping digital well being adoption, a number of the regulatory shifts underway in digital well being and the way BrightInsight is collaborating with firms to satisfy this new imaginative and prescient of healthcare.
One deal that underscores this pattern is the digital well being collaboration between Fitbit, owned by Google, and Verily’s digital clinic – Onduo. The collaboration pairs Onduo’s continual situation platform with Fitbit’s units and Fitbit Premium as a part of the corporate’s method to complete individual well being. It seeks to offer a complete view of how the totally different facets of well being and wellbeing match collectively, together with exercise, sleep and stress, to assist members higher handle their well being and construct a routine that works for them, based on the September announcement.
These sorts of collaborations are additionally going down in life sciences, for a similar causes.
“How do you leverage steady knowledge sources in ways in which drive intervention with the affected person that transcend a affected person interplay having to be a head to head medical go to?” Patel requested.
“Our complete portfolio speaks to the potential and the facility of what you are able to do by wrapping regulated software program round conventional medicine and units.”
Digital well being funding soared to new heights in 2021, significantly for firms growing digital care tech. As an organization that gives the infrastructure for pharma and medtech firms to develop and scale regulated digital well being packages, BrightInsight benefited from this funding pattern as nicely. Within the first quarter, it closed a $101 million Sequence C spherical led by Normal Catalyst.
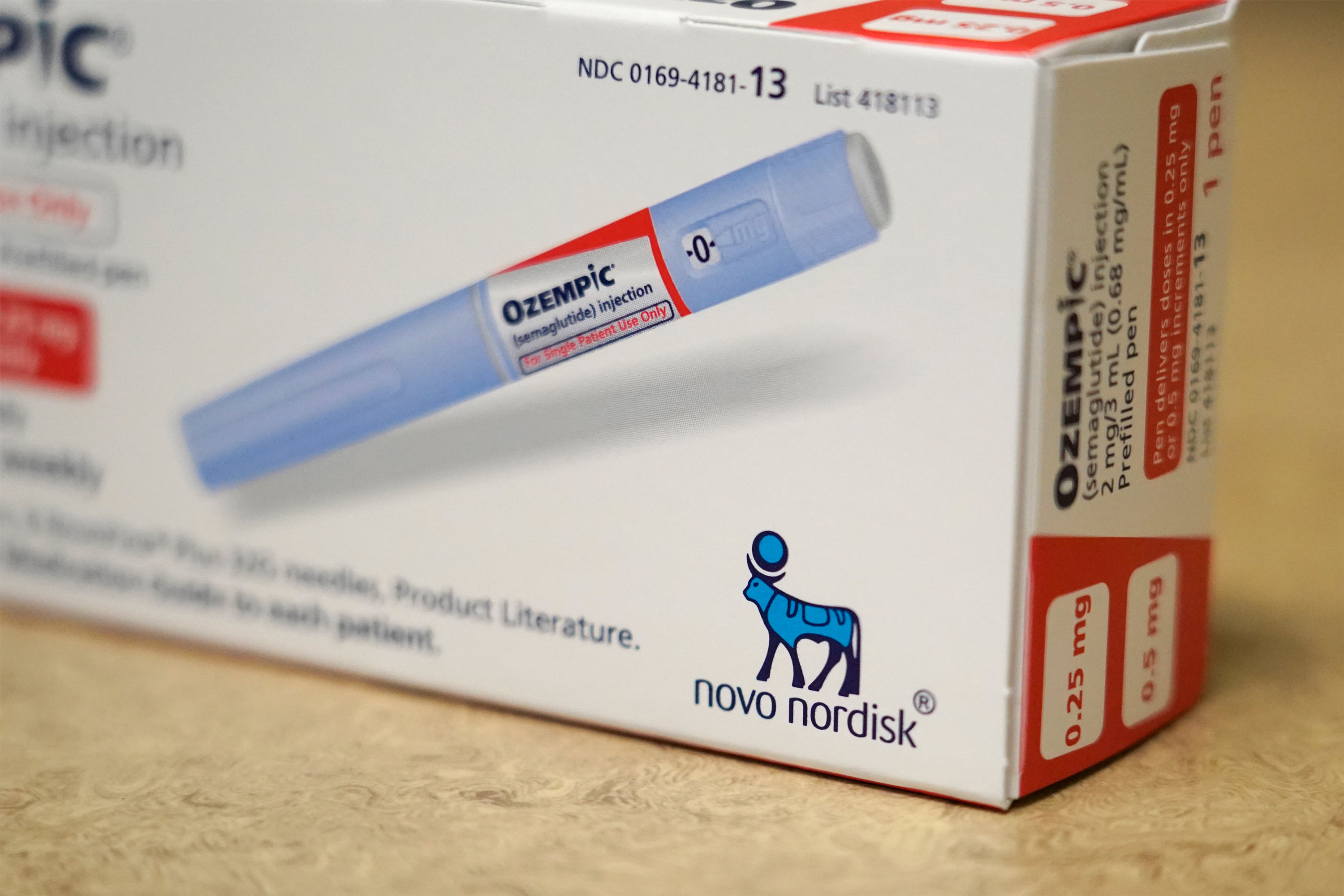
Patel’s background in digital well being in pharma at Amgen, with telemedicine supplier Physician on Demand and newer expertise as president of Flex Digital Well being makes him nicely suited to steer these collaborations. One instance of how BrightInsight helps life science firms execute on their digital care ambitions is its collaboration with Novo Nordisk to assist folks with Sort 2 diabetes.
The pharma firm wanted a regulated digital well being platform to assist it construct and function a set of digital well being services to enhance diabetes care. Along with an open, device-agnostic platform that might seize knowledge from steady blood glucose meters and insulin pens, Novo Nordisk required a regulated platform designed to fulfill the stringent privateness, safety, regulatory and high quality necessities of US and European healthcare regulatory our bodies. One of many challenges that firms search to deal with is to enhance the best way sufferers handle their continual circumstances. At the least 45% of individuals with sort 2 diabetes have a tricky time managing their blood sugar ranges and drugs adherence is one contributing issue.
The collaboration with BrightInsight helps folks with diabetes unlock actual world knowledge. By integrating consumer’s blood glucose knowledge with robotically recorded insulin dose knowledge from Novo Nordisk’s good insulin pens, customers can higher perceive how their actions, food plan, and insulin consumption play a job of their ranges and make extra knowledgeable selections to maintain their glucose ranges inside protected parameters.
Though there are quite a few firms growing digital well being options, if they’ll’t adapt to regulatory shifts, they danger falling behind or changing into irrelevant. Earlier this yr, the EU regulatory physique mandated a collection of adjustments directing how firms must handle software program medical units, which went into impact Might 26. The adjustments have been designed to make sure a excessive customary of security and high quality as regulators attempt to sustain with the tempo of well being tech innovation whereas sustaining the necessity for security and efficacy. The FDA shared draft steerage for deliberate adjustments to premarket submission of digital well being expertise – the unique model dates again to 2005.
The FDA has indicated that it’s updating software program medical system steerage so that every software program system will fall into one in every of two buckets: fundamental or enhanced, based on a weblog publish on BrightInsight’s web site. The up to date draft steerage is the FDA’s try to sustain with the inflow of cutting-edge submissions. By specializing in fundamental and enhanced danger, they eradicate the center floor and streamline the submission course of.
“That is reflective of the FDA’s view of low danger/excessive danger in relation to units,” mentioned Sonia Nath, a accomplice with Cooley legislation agency in a BrightInsight weblog publish. “This can be introduced as a less complicated risk-based method to viewing software program units usually…The considering behind this steerage is, if we let you know extra explicitly what we want, you will get via the approval course of extra shortly as a result of there’s not going to be a lot forwards and backwards. Expertise is at all times quicker than the legislation and the steerage paperwork.”
The brand new draft pointers would require that submissions for “excessive danger” software program units are accompanied by extra design documentation spelling out the technical design particulars of how the software program capabilities, how the software program design implements all the necessities of the Software program Requirement Specification (SRS) and the way the Software program Design Specification (SDS) traces to the SRS when it comes to meant use, performance, security, and effectiveness.
The convergence of pharma, medtech and suppliers via digital well being knowledge over the previous few years has been an interesting transformation. Pharma firms resembling Takeda and Sanofi are growing specialty practices to achieve larger insights on market dynamics and what sufferers want. Patel famous that well being knowledge has turn out to be a important commodity.
“For pharma and medical system firms, there’ll proceed to be an incredible growth and focus of their assets going into software program,” Patel mentioned. “You’ll have a number of sources of well being knowledge that may reply questions from illness analysis to matching sufferers with the precise therapies.”
He added: “I feel all illness areas are amenable to digital options.”
Digital well being visits are poised to extend in 2022 as will the necessity for regulated related units and Software program as a Medical Gadget (SaMD) to watch affected person well being between medical appointments. Gathering and analyzing knowledge from these units shouldn’t be solely important for recognizing potential issues with dosages, system utilization and the general well being of sufferers; they’ll additionally make the interplay between sufferers and their clinicians extra significant with the insights they’ll present to them. The digital well being choices throughout pharma and medtech will proceed to develop and turn out to be extra sturdy throughout continual circumstances and specialised areas resembling oncology and uncommon illnesses. Will probably be attention-grabbing to see the shape these collaborations take within the new yr.
Photograph: NicoElNinom, Getty Photographs