Ring-shaped chemical buildings known as saturated heterocycles are present in most FDA-approved medication however are sometimes tough to create. Scripps Analysis chemists have simply developed a surprisingly simple methodology for making many of those sought-after compounds from cheap beginning chemical substances.
The brand new methodology, described in a paper that seems April 11, 2024, in Nature Synthesis, permits chemists to make saturated heterocycles from comparatively easy, chain-like amine compounds.
The researchers demonstrated the ability of their new methodology through the use of it to carry out an environment friendly synthesis of stemoamide, a fancy plant-derived compound present in conventional medicines.
“These new reactions ought to make it simpler than ever to assemble saturated heterocycles with ring sizes and buildings which can be related for drug improvement,” says research senior writer Jin-Quan Yu, Ph.D., Frank and Bertha Hupp Professor of Chemistry and Bristol Myers Squibb Endowed Chair in Chemistry at Scripps Analysis.
The primary writer was Sam Chan, Ph.D., a postdoctoral analysis affiliate within the Yu lab in the course of the research.
Saturated heterocycles are cyclic natural compounds whose spine construction comprises no less than one non-carbon atom. In heterocyclic drug compounds, the non-carbon atom is normally a nitrogen atom, which regularly performs a vital function in figuring out the compound’s chemical properties and therapeutic effectiveness. Nevertheless, present strategies for making these much-valued compounds are fairly restricted. Even after they can be utilized, they are typically cumbersome or require comparatively costly and sophisticated beginning supplies.
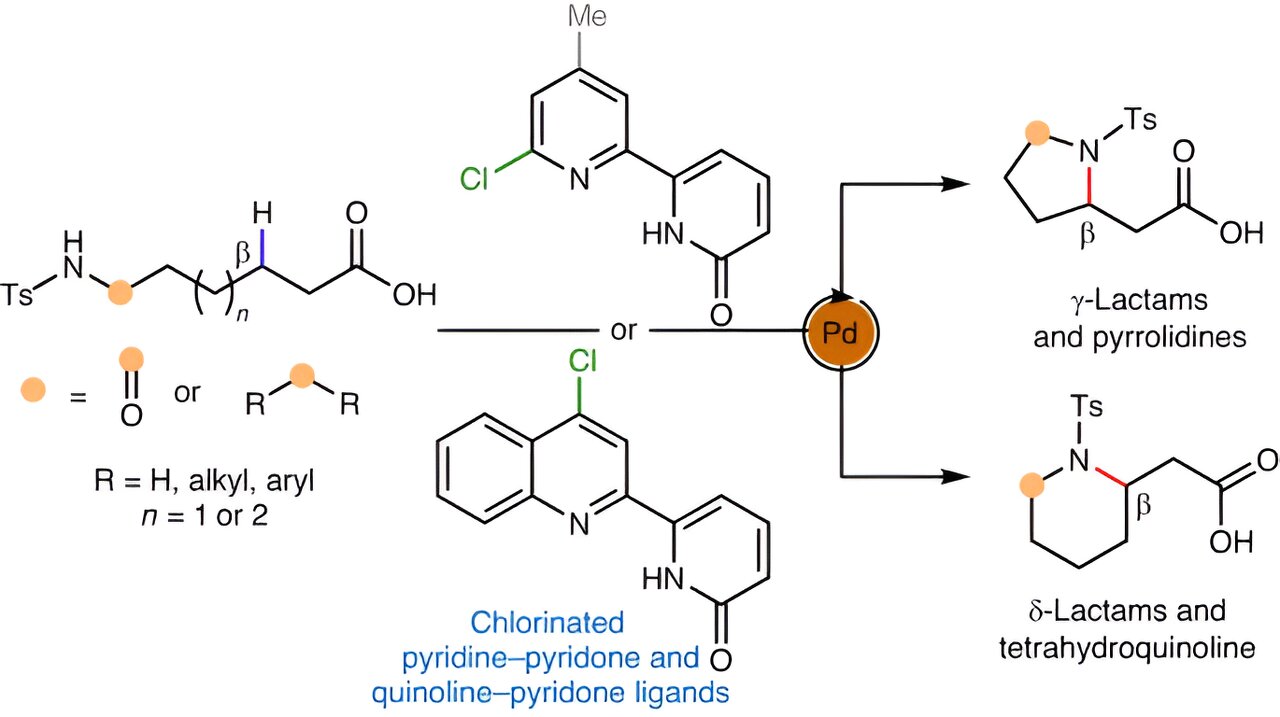
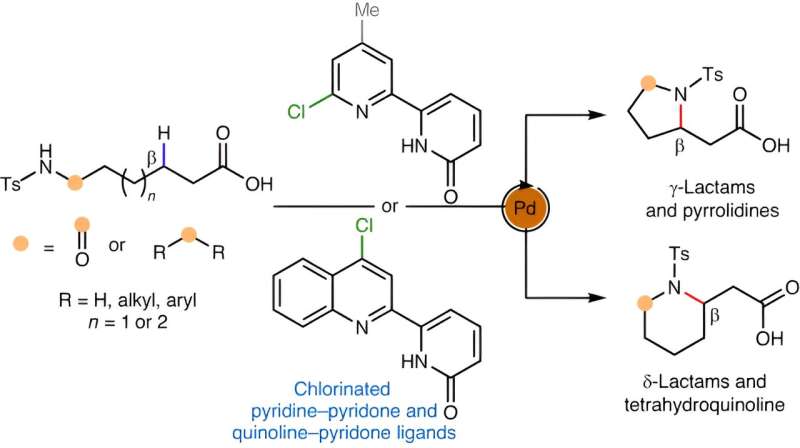
“Probably the most handy solution to forge such a hoop could be to take a available aliphatic amine compound, which comprises nitrogen, and sew that nitrogen onto one other a part of its carbon spine, basically folding the molecule onto itself,” Yu says.
This could contain the elimination of a hydrogen atom to permit the brand new carbon-nitrogen bond to kind—making it a sort of “C-H activation” response, lengthy the specialty of the Yu lab. No such C-H response for forming cyclic amines has existed—not a sensible one, anyway. For the brand new research, Yu and his staff got down to invent one.
The strategy they finally devised included a palladium catalyst for breaking the C-H bond. It additionally concerned a set of molecules known as chlorinated pyridine-pyridones that work as so-called ligands with the right geometry to advertise the brand new C-N bond formation.
The chemists showcased their new strategy by simply making dozens of cyclic amine and associated buildings, together with γ- and δ-lactams, pyrrolidines, and tetrahydroquinolines—all of which might be of curiosity to pharmaceutical chemists.
In a ultimate flourish, they showcased the utility of their methodology with a synthesis—beginning nearly from scratch with a quite simple amine compound—of the plant-derived cyclic amide stemoamide, which has been considered as a possible start line for brand spanking new anti-inflammatory medication.
Yu and his staff are at present working to increase their new strategy to make different varieties of saturated heterocycle.
“Palladium-catalyzed methylene C(sp3)–H lactamization and cycloamination enabled by chlorinated pyridine-pyridone ligands” was co-authored by Hau Solar Sam Chan, Yilin Lu, and Jin-Quan Yu, all of Scripps Analysis.
Extra data:
Hau Solar Sam Chan et al, Palladium-catalysed methylene C(sp3)–H lactamization and cycloamination enabled by chlorinated pyridine–pyridone ligands, Nature Synthesis (2024). DOI: 10.1038/s44160-024-00517-5
Supplied by
The Scripps Analysis Institute
Quotation:
Chemists devise simpler new methodology for making a standard sort of constructing block for medication (2024, April 13)
retrieved 14 April 2024
from https://phys.org/information/2024-04-chemists-easier-method-common-block.html
This doc is topic to copyright. Other than any truthful dealing for the aim of personal research or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for data functions solely.