Impressed by what human liver enzymes can do, Scripps Analysis chemists have developed a brand new set of copper-catalyzed natural synthesis reactions for constructing and modifying prescription drugs and different molecules. The brand new reactions are anticipated to be extensively utilized in drug discovery and optimization, in addition to in different chemistry-based industries.
Of their examine, printed in Nature, the chemists confirmed that their new strategies can be utilized to carry out two modifications—referred to as dehydrogenations and lactonizations—on a broad class of cheap beginning compounds. The reactions require solely a easy copper-based catalyst, whereas associated reactions usually require way more cumbersome and costly strategies—although this particular kind of response was beforehand inaccessible by any natural synthesis technique.
“This new two-mode method might be significantly helpful for late-stage modifications and variations of pure merchandise and drug molecules,” says examine senior writer Jin-Quan Yu, Ph.D., Frank and Bertha Hupp Professor of Chemistry and Bristol Myers Squibb Endowed Chair in Chemistry at Scripps Analysis.
The examine’s first authors have been postdoctoral analysis affiliate Shupeng Zhou, Ph.D., and doctoral pupil Annabel Zhang, Ph.D., each of the Yu lab through the examine.
The preliminary objective of the analysis was to discover a new and higher technique for what chemists name carbon-hydrogen (CH) activation, by which a hydrogen atom on the carbon spine of an natural compound is indifferent and changed with one thing else—a invaluable instrument for drug synthesis.
On this case, the Yu lab—which has a historical past of improvements in CH activation chemistry—sought a greater solution to do CH activations that change the hydrogen with an oxygen atom. This can be a widespread transformation within the development or modification of biologically energetic molecules, although chemists have not had laboratory strategies for doing it which are as easy, direct and broadly helpful as they want.
Yu and his workforce appeared to nature for inspiration, particularly to cytochrome P450 enzymes, that are present in most residing organisms, and assist clear doubtlessly poisonous molecules within the human liver. Cytochrome P450 enzymes carry out oxygen-for-hydrogen reactions very effectively.
A few of these enzymes have the extra capability to catalyze a special hydrogen-removal course of referred to as dehydrogenation, which can be utilized to strip hydrogens from two carbons concurrently, permitting different atoms—or clusters of atoms—to interchange them. The chemists set themselves the bold objective of discovering a normal natural synthesis technique for doing both the oxygenation or dehydrogenation response, as these versatile “bimodal” enzymes do in residing cells.
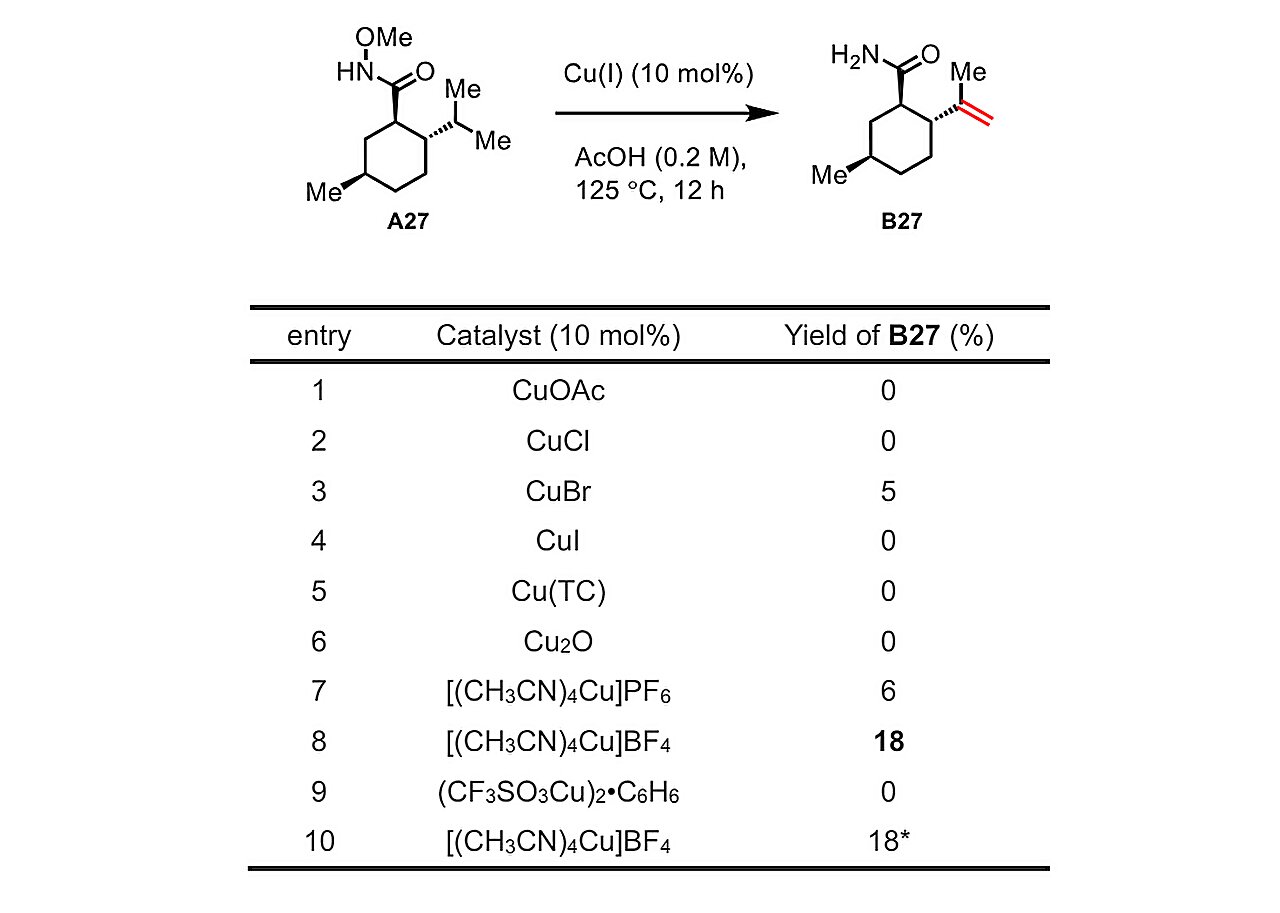
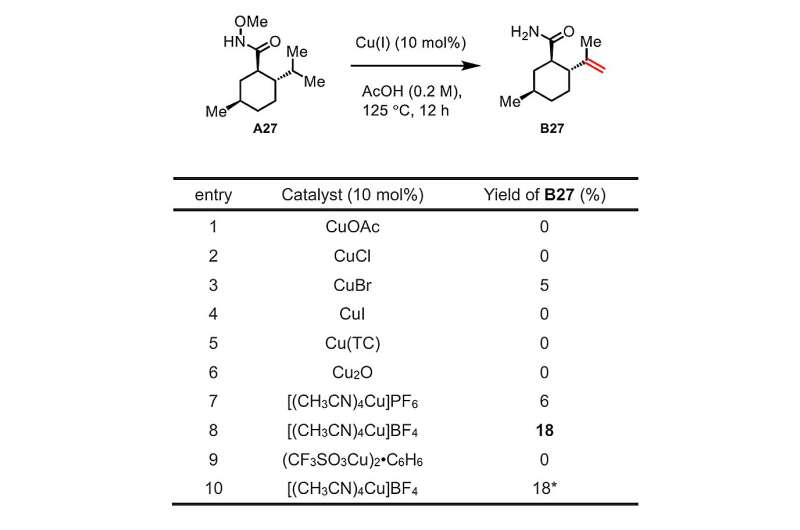
After months of experimentation, Yu’s workforce discovered that, via chemical transformations just like these finished by the bimodal cytochrome P450 enzymes, they might effectively make compounds referred to as unsaturated major amides—a category that features many drug molecules—by dehydrogenating cheap beginning compounds referred to as methoxyamides. For the catalyst, they wanted solely copper fluoride—additionally cheap and straightforward to make use of.
Because the chemists explored the breadth of their new dehydrogenation technique utilizing completely different particular beginning compounds, they noticed hint quantities of a sort of molecule referred to as a lactone, indicating that an oxygenation response had occurred. In the end, they have been capable of decide the response circumstances that favored this oxygenation or “lactonization” over the dehydrogenation. In different phrases, just like the bimodal enzymes that had impressed them, they have been capable of management whether or not their method led down one response path or the opposite.
The workforce demonstrated the outstanding versatility of this set of reactions through the use of it to change—by way of dehydrogenation or lactonization, or each—all kinds of beginning compounds, together with the neurological drug valproic acid and the cholesterol-lowering drug gemfibrozil. (Modifications of present advanced molecules to create doubtlessly higher variants are a typical drug discovery and optimization approach.)
Yu and his group are presently growing the same method for making and modifying lactone- and amide-related compounds referred to as lactams, which embrace some antibiotics.
“We have already had a variety of curiosity on this new method from pharma business chemists,” Yu says.
“Copper-catalyzed dehydrogenation or lactonization of C(sp3)−H bonds” was co-authored by Shupeng Zhou, Zi-Jun Zhang and Jin-Quan Yu.
Extra info:
Shupeng Zhou et al, Copper-catalyzed dehydrogenation or lactonization of C(sp3)−H bonds, Nature (2024). DOI: 10.1038/s41586-024-07341-z
Offered by
The Scripps Analysis Institute
Quotation:
Chemists introduce new copper-catalyzed C-H activation technique (2024, April 19)
retrieved 19 April 2024
from https://phys.org/information/2024-04-chemists-copper-catalyzed-strategy.html
This doc is topic to copyright. Aside from any truthful dealing for the aim of personal examine or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for info functions solely.