Researchers on the College of Pittsburgh and Drexel College in Philadelphia, together with Brookhaven Nationwide Laboratory, are working to resolve a multipart thriller to make water disinfection remedies extra sustainable.
Scalable electrochemical ozone manufacturing (EOP) applied sciences to disinfect soiled water might sometime exchange centralized chlorine remedies used immediately, whether or not in trendy cities or distant villages. Nonetheless, little is known about EOP on the molecular degree and the way applied sciences that make it attainable might be made to be environment friendly, economical, and sustainable.
Their analysis, “Interaction between Catalyst Corrosion and Homogeneous Reactive Oxygen Species in Electrochemical Ozone Manufacturing,” was printed not too long ago within the journal ACS Catalysis.
The lead creator is Drexel Ph.D. pupil Rayan Alaufey, with contributing researchers from Drexel, together with co-PI Maureen Tang, affiliate professor of chemical and organic engineering, postdoctoral researcher Andrew Lindsay, Ph.D. pupil Tana Siboonruang, and Ezra Wooden, affiliate professor of chemistry; co-PI John A. Keith, affiliate professor of chemical and petroleum engineering, and graduate pupil Lingyan Zhao from Pitt; and Qin Wu from Brookhaven.
“Individuals have used chlorine to deal with ingesting water for the reason that nineteenth century, however immediately we higher perceive that chlorine might not all the time be the best choice. EOP for instance can generate ozone, a molecule with about the identical disinfecting energy as chlorine, immediately in water.
“In contrast to chlorine which stably persists in water, ozone in water naturally decomposes after about 20 minutes, which means it’s much less more likely to injury folks when consuming from water at a faucet, when swimming in a pool, or when cleansing wounds in a hospital,” defined Keith, who can also be R.Okay. Mellon College Fellow in Power at Pitt’s Swanson College of Engineering.
“EOP for sustainable disinfection would make lots of sense in some markets, however doing it requires a adequate catalyst, and since no one has discovered a adequate EOP catalyst but, EOP is simply too costly and energy-intensive for broader use.
“My colleagues and I assumed if we may decode on the atomic degree what makes a mediocre EOP catalyst work, possibly we may engineer an excellent higher EOP catalyst.”
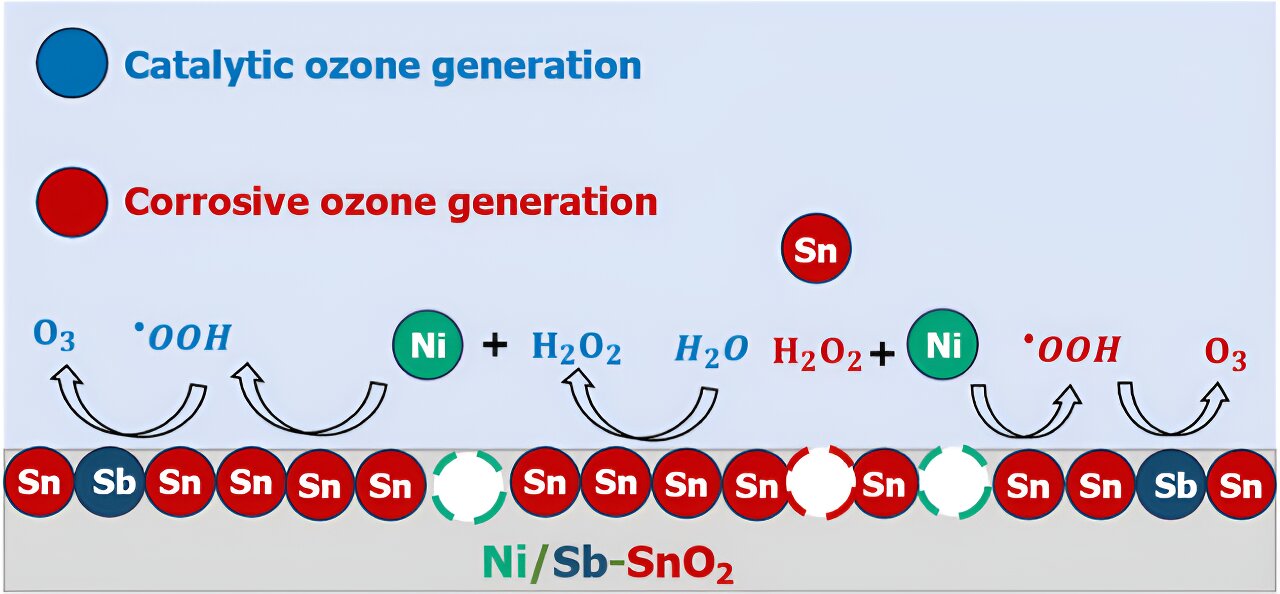
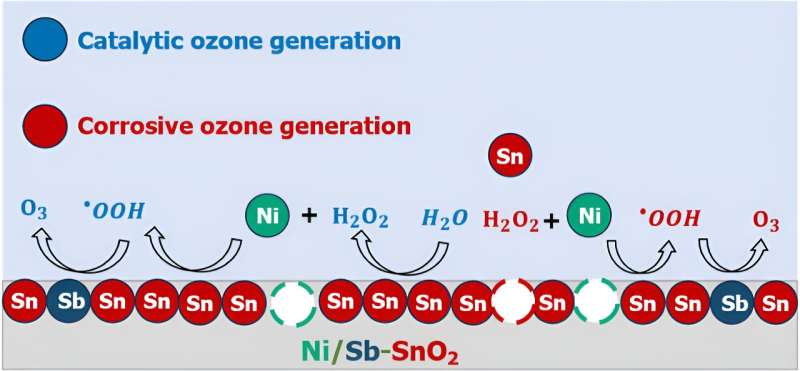
Fixing the thriller of how EOP catalysts work is essential in understanding find out how to higher engineer one of the promising and least poisonous EOP catalysts identified up to now: nickel- and antimony-doped tin oxide (Ni/Sb–SnO2, or NATO).
Therein, mentioned Keith, lies the conundrum: what’s each atom’s function in NATO doing to assist EOP? Is ozone getting shaped catalytically in methods we wish it to, or does it kind as a result of the catalyst is decomposing, and future work must be finished to make NATO catalysts extra steady?
Surprisingly, the researchers found that it’s in all probability a mixture of each.
By utilizing experimental electrochemical analyses, mass spectrometry, and computational quantum chemistry modeling, the researchers created an “atomic-scale storyline” to elucidate how ozone is generated on NATO electrocatalysts.
For the primary time, they recognized that among the nickel in NATO might be leaching out of the electrodes through corrosion, and these nickel atoms, now floating within the answer close to the catalyst, can promote chemical reactions that finally generate ozone.
“If we need to make a greater electrocatalyst, we have to perceive what elements are working and never working. Components like metallic ion leaching, corrosion, and answer section reactions can provide the looks {that a} catalyst is working a method when truly it’s working one other manner.”
Keith famous that figuring out the prevalence of corrosion and chemical reactions occurring away from the catalyst are necessary steps to make clear earlier than different researchers can pursue enhancements to EOP and different electrocatalytic processes.
Of their conclusion, they notice, “Figuring out or refuting the existence of such basic technological constraints might be important to any future functions of EOP and different superior electrochemical oxidation processes.”
“We all know that electrochemical water remedy works on small scales, however the discovery of higher catalysts will increase it to a worldwide scale. The following step is discovering new atomic combos in supplies which can be extra immune to corrosion but additionally promote economically and sustainably viable EOP,” Keith mentioned.
Extra info:
Rayan Alaufey et al, Interaction between Catalyst Corrosion and Homogeneous Reactive Oxygen Species in Electrochemical Ozone Manufacturing, ACS Catalysis (2024). DOI: 10.1021/acscatal.4c01317
Offered by
College of Pittsburgh
Quotation:
Engineers clear up ‘catalysis vs corrosion’ thriller in electrochemical ozone manufacturing (2024, Might 6)
retrieved 7 Might 2024
from https://phys.org/information/2024-05-catalysis-corrosion-mystery-electrochemical-ozone.html
This doc is topic to copyright. Other than any truthful dealing for the aim of personal examine or analysis, no
half could also be reproduced with out the written permission. The content material is offered for info functions solely.