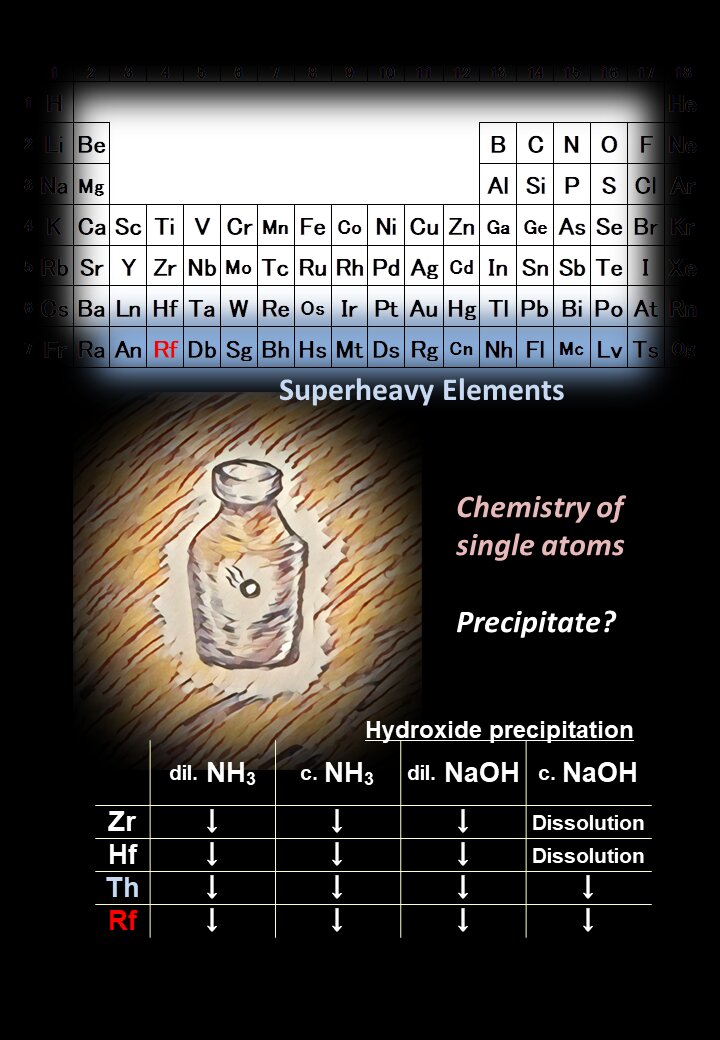
All chemistry college students are taught in regards to the periodic desk, a company of the weather that helps you establish and predict traits of their properties. For instance, science fiction writers typically describe life primarily based on the component silicon as a result of it’s in the identical column within the periodic desk as carbon.
Nevertheless, there are deviations from anticipated periodic traits. For instance, lead and tin are in the identical column within the periodic desk and thus ought to have comparable properties. Nevertheless, while lead-acid batteries are widespread in vehicles, tin-acid batteries do not work. These days we all know that it’s because a lot of the vitality in lead-acid batteries is attributable to relativistic chemistry however such chemistry was unknown to the researchers who initially proposed the periodic desk.
Relativistic chemistry is troublesome to review within the superheavy parts, as a result of such parts are usually produced one after the other in nuclear fission reactions and deteriorate shortly. However, being able to review the chemistry of superheavy parts might uncover new purposes for superheavy parts and for widespread lighter parts, comparable to lead and gold.
In a latest examine in Nature Chemistry, researchers from Osaka College studied how single atoms of superheavy rutherfordium steel react with two lessons of widespread bases. Such experiments will assist researchers use relativistic ideas to higher make the most of the chemistry of many parts.
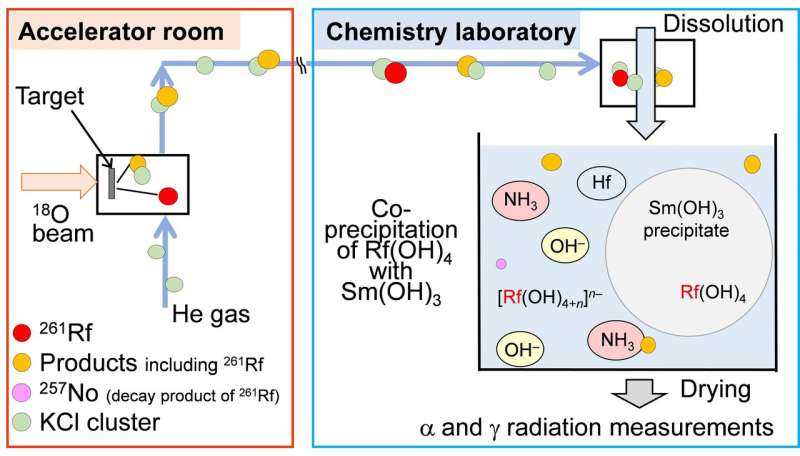
“We ready single atoms of rutherfordium at RIKEN accelerator analysis facility, and tried to react these atoms with both hydroxide bases or amine bases,” explains Yoshitaka Kasamatsu, lead writer on the examine. “Radioactivity measurements indicated the top end result.”
Researchers can higher perceive relativistic chemistry from such experiments. For instance, rutherfordium varieties precipitate compounds with hydroxide base in any respect concentrations of base, but its homologues zirconium and hafnium in excessive concentrations. This distinction in reactivity could also be attributable to relativistic chemistry.
“If we had a strategy to produce a pure rutherfordium precipitate in bigger portions, we might transfer ahead with proposing sensible purposes,” says senior writer Atsushi Shinohara. “Within the meantime, our research will assist researchers systematically discover the chemistry of superheavy parts.”
Relativistic chemistry explains why bulk gold steel isn’t silver-colored, as one would anticipate primarily based on periodic desk predictions. Such chemistry additionally explains why mercury steel is a liquid at room temperature, regardless of periodic desk predictions. There could also be many unexpected purposes that come up from studying in regards to the chemistry of superheavy parts. These discoveries will depend upon newly reported protocols and ongoing elementary research comparable to this one by Osaka College researchers.
Superheroes, meals and apps carry a contemporary twist to the periodic desk
Co-precipitation behaviour of single atoms of rutherfordium in primary options. Nature Chemistry. DOI: 10.1038/s41557-020-00634-6
Osaka College
Quotation:
Experimental checks of relativistic chemistry will replace the periodic desk (2021, February 16)
retrieved 16 February 2021
from https://phys.org/information/2021-02-experimental-relativistic-chemistry-periodic-table.html
This doc is topic to copyright. Other than any truthful dealing for the aim of personal examine or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for info functions solely.