A pair of coronary heart gadgets linked to lots of of accidents and no less than 14 deaths has acquired the FDA’s most severe recall, the company introduced Monday.
The recall comes years after surgeons say they first seen issues with the HeartMate II and HeartMate 3, manufactured by Thoratec Corp., a subsidiary of Abbott Laboratories. The gadgets usually are not at the moment being faraway from the market. In an emailed response, Abbott mentioned it had communicated the chance to prospects this 12 months.
The delayed motion raises questions for some security advocates about how and when points with authorized medical gadgets must be reported. The guts gadgets in query have been related to hundreds of stories of sufferers’ accidents and deaths, as described in a KFF Well being Information investigation late final 12 months.
“Why doesn’t the general public know?” mentioned Sanket Dhruva, a heart specialist and an knowledgeable in medical machine security and regulation on the College of California-San Francisco. Although some surgeons could have been conscious of points, others, significantly those that don’t implant the machine ceaselessly, could have been in the dead of night. “And their sufferers are struggling opposed occasions,” he mentioned.
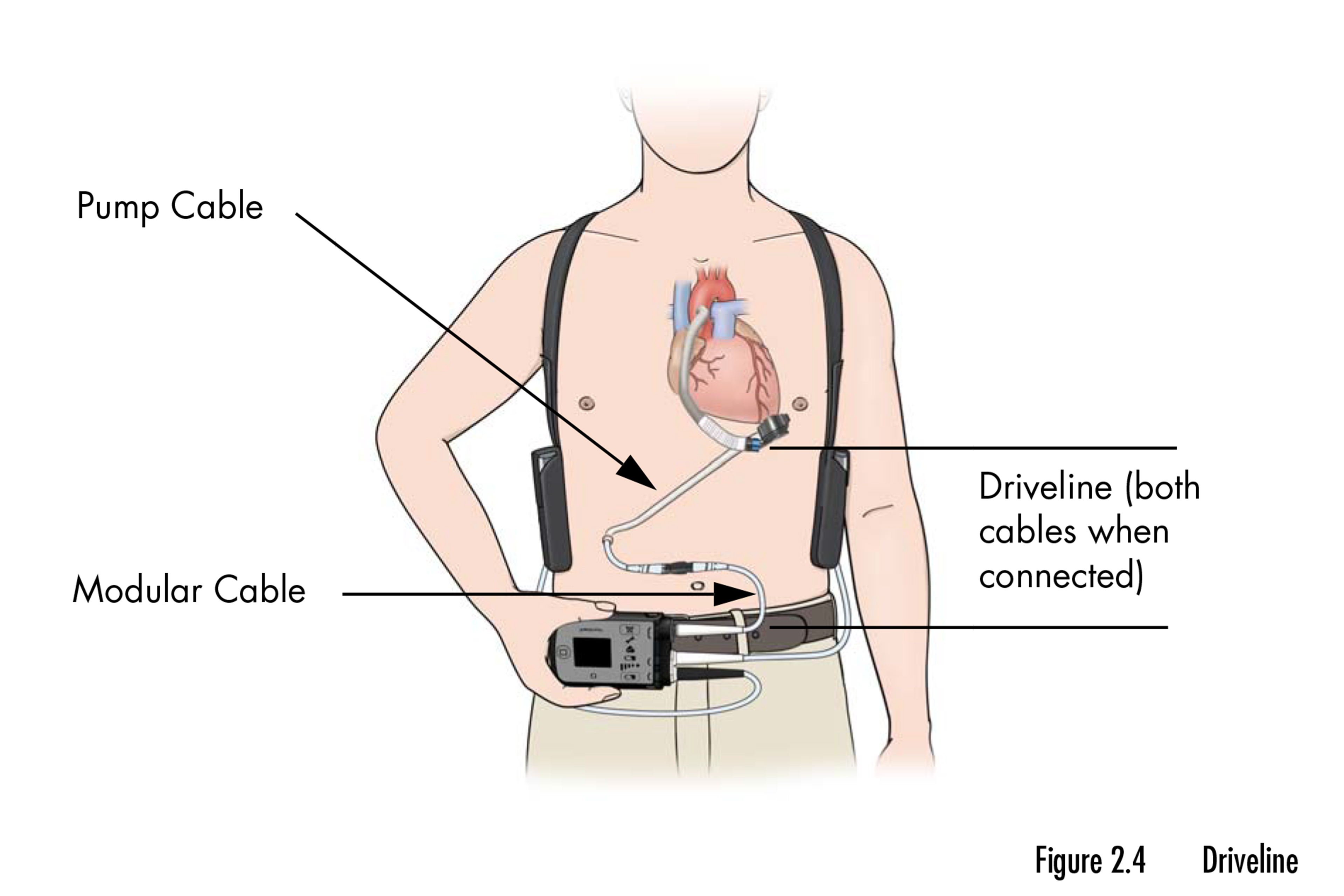
The recall entails a pair of mechanical pumps that assist the guts pump blood when it will probably’t accomplish that by itself. The gadgets, sufficiently small to slot in the palm of a hand, are implanted in sufferers with end-stage coronary heart failure who’re ready for a transplant or as a everlasting resolution when a transplant shouldn’t be an choice. The recall impacts almost 14,000 gadgets.
Amanda Hils, an FDA press officer, mentioned the company is working with Abbott to research the reported accidents and deaths and decide if additional motion is required.
“So far, the variety of deaths reported seems in keeping with the opposed occasions noticed within the preliminary scientific trial,” Hils mentioned in an e-mail.
In response to the FDA’s recall discover, the gadgets may cause buildup of “organic materials” that reduces their means to assist the guts flow into blood and preserve sufferers alive. The buildup accumulates step by step and may seem two years or extra after a tool is implanted in a affected person’s chest.
Docs had been suggested to be careful for “low-flow alarms” on the gadgets and, in the event that they do diagnose the obstruction, to both monitor the affected person or carry out surgical procedure to implant a stent, launch the blockage, or exchange the pump. “Charges of outflow obstruction are low,” Abbott spokesperson Justin Paquette mentioned in an e-mail, including that sufferers whose LVADs are functioning usually “don’t have any purpose for concern.”
A assessment of the FDA machine database exhibits no less than 130 stories associated to HeartMate II or 3 that point out the complication reported by regulators. The earliest such report filed with the FDA dates to no less than 2020, based on a KFF Well being Information assessment of the database.
Monday’s alert is the second Class 1 recall of a HeartMate machine this 12 months.
In January, Abbott issued an pressing “correction letter” to hospitals a few separate problem during which the HeartMate 3 unintentionally begins and stops because of the pump’s communication system, which cardiologists use to evaluate sufferers’ standing. The FDA alerted the general public in March.
In February, Abbott issued one other pressing letter to hospitals in regards to the blockage drawback, asking them to tell physicians, full and return an acknowledgment kind, and take note of low-flow alarms on the machine’s monitor that will point out an obstruction. The corporate mentioned within the letter that it’s engaged on “a design resolution” to stop the blockages.
A examine printed in 2022 within the Journal of Thoracic and Cardiovascular Surgical procedure reported the obstruction in about 3% of circumstances, although the incidence fee was increased the longer a affected person had the machine.
The one different Class 1 recall issued for the HeartMate 3 was in Could 2018, when the corporate issued corrective motion notices to hospitals and physicians warning that the graft line that carries blood from the pump to the aorta might twist and cease blood stream.
The FDA recall discover issued Monday contains further steerage for physicians to diagnose the blockage utilizing an algorithm to detect obstructions and, if wanted, a CT angiogram to confirm the trigger.
At current, the HeartMate 3, which was first authorized by the FDA in 2017, is the one medical choice for a lot of sufferers with end-stage coronary heart failure and who don’t qualify for a transplant. The HeartMate 3 has supplanted the HeartMate II, which acquired FDA approval in 2008.
If the brand new recall results in the machine being faraway from the market, end-stage coronary heart failure sufferers might don’t have any choices, mentioned Francis Pagani, a cardiothoracic surgeon on the College of Michigan who additionally oversees a proprietary database of HeartMate II and HeartMate 3 implants.
If that occurs, “we’re in bother,” Pagani mentioned. “It will be devastating to the sufferers to not have this selection. It’s not an ideal choice — no pump ever is — however that is nearly as good because it’s ever been.”
It’s not identified exactly what number of sufferers have acquired a HeartMate II or HeartMate 3 implant. That data is proprietary. The FDA recall notices present worldwide distribution of greater than 22,000 HeartMate 3 gadgets and greater than 2,200 of the HeartMate II.
The blockage complication could have gone unreported to the general public for thus lengthy partly as a result of physicians usually are not required to report opposed occasions to federal regulators, mentioned Madris Kinard, a former FDA medical machine official and founding father of Gadget Occasions, an organization that makes FDA machine information extra user-friendly for hospitals, regulation companies, and traders.
Solely machine producers, machine importers, and hospitals are required by regulation to report device-related accidents, deaths, and important malfunctions to the FDA.
“If that is one thing physicians had been conscious of, however they weren’t mandated to report back to the FDA,” Kinard mentioned, “at what level does that communication between these two teams have to occur?”
Dhruva, the heart specialist, mentioned he’s in search of transparency from Abbott about what the corporate is doing to handle the issue so he can have extra thorough conversations with sufferers contemplating a HeartMate machine.
“We’re going to anticipate to have some information saying, ‘Hey we created this repair, and this repair works, and it doesn’t trigger a brand new drawback.’ That’s what I need to know,” he mentioned. “There’s only a ton extra that I really feel in the dead of night about, to be trustworthy, and I’m positive that sufferers and their households do as effectively.”
[Update: This article was updated at 5:20 p.m. ET on April 16, 2024, with a response from Abbott Laboratories, which it provided after publication.]