Scientific trials have detailed examine protocols and are registered on ClinicalTrials.gov. What degree of particulars are wanted for real-world information (RWD) analyses that goal to estimate therapy results? In Europe, the European Medicines Company (EMA) requires registration of many examine protocols utilizing a template for observational post-authorization security research (PASS) carried out by advertising and marketing authorization holders. Different efforts embody ISPE’s tips for good pharmacoepidemiology observe (GPP) part on protocol improvement, Nationwide Analysis System for well being Expertise (NEST) protocol steerage, and the Structured Template and Reporting Device for Actual World Proof (STaRT-RWE).
The Worldwide Society for Pharmacoepidemiology (ISPE) and the Worldwide Society for Pharmacoeconomics and Outcomes Analysis (ISPOR) convened a joint process power to supply requirements for RWD protocols. They developed the HARmonized Protocol Template to Improve Reproducibility (HARPER). The Desk under compares HARPER towards PASS, GPP, NEST and STaRT-RWE.
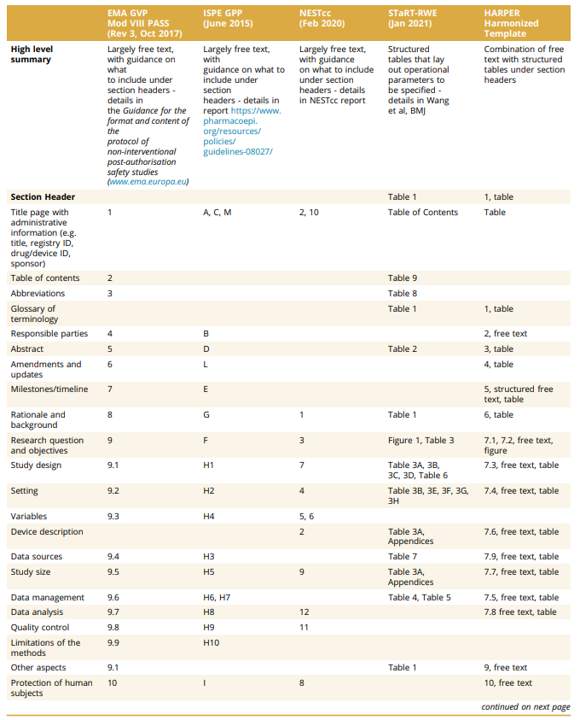

The authors correctly notice a number of limitations of HARPER.
- Structured vs. Flexibility. HARPER’s structured strategy enhances readability for stakeholders and consistency throughout research. Nevertheless, some advanced examine designs could also be fully affordable, however could not match inside the HARPER construction. Most of the sections, nonetheless, comprise free textual content sections.
- Minimal, not most transparency. HARPER doesn’t cowl each facet of transparency over the lifecycle of a analysis examine, which can contain sharing of protocol, code, information, in addition to outcomes. Thus, HARPER needs to be seen because the minimal necessities for examine protocol transparency.
- Knowledge evolution. As information assortment evolves and new strategies are developed, HARPER’s strategy could have to adapt over time.
The complete HARPER Desk of Contents is listed under.
- 1. Title Web page
- 2. Summary
- 3. Amendments and updates
- 4. Timeline
- Desk 1 Milestones and Timeline
- 5. Rationale and background
- 6. Analysis query and goals
- Desk 2 Main and secondary analysis questions and goal
- 7. Analysis strategies
- 7.1. Examine design
- 7.2. Examine design diagram
- 7.3. Setting
- 7.3.1 Context and rationale for definition of time 0 (and different major time anchors) for entry to the examine inhabitants
- Desk 3 Operational Definition of Time 0 (index date) and different major time anchors
- 7.3.2 Context and rationale for examine inclusion standards:
- Desk 4. Operational Definitions of Inclusion Standards
- 7.3.3 Context and rationale for examine exclusion standards
- Desk 5. Operational Definitions of Exclusion Standards
- 7.3.1 Context and rationale for definition of time 0 (and different major time anchors) for entry to the examine inhabitants
- 7.4. Variables
- 7.4.1 Context and rationale for publicity(s) of curiosity
- Desk 6. Operational Definitions of Publicity
- 7.4.2 Context and rationale for final result(s) of curiosity
- Desk 7. Operational Definitions of End result
- 7.4.3 Context and rationale for observe up
- Desk 8. Operational Definitions of Observe-Up
- 7.4.4 Context and rationale for covariates (confounding variables and impact modifiers, e.g. danger elements, comorbidities, comedications)
- Desk 9. Operational Definitions of Covariates
- 7.4.1 Context and rationale for publicity(s) of curiosity
- 7.5. Knowledge evaluation
- 7.5.1 Context and rationale for evaluation plan
- Desk 10. Main, secondary, and subgroup evaluation specification
- Desk 11. Sensitivity analyses – rationale, strengths and limitations
- 7.5.1 Context and rationale for evaluation plan
- 7.6. Knowledge sources
- 7.6.1 Context and rationale for information sources
- Desk 12. Metadata about information sources and software program
- 7.6.1 Context and rationale for information sources
- 7.7. Knowledge administration
- 7.8. High quality management
- 7.9. Examine dimension and feasibility
- Desk 13. Energy and pattern dimension
- 8. Limitation of the strategies
- 9. Safety of human topics
- 10. Reporting of adversarial occasions
- 11. References
- 12. Appendices
You may learn extra particulars on the HARPER strategy right here and on the Open Science Basis right here.






