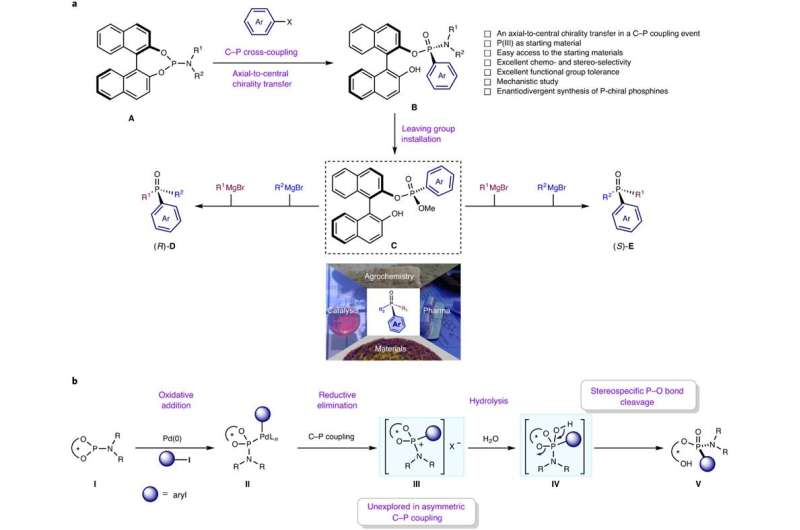
Chiral phosphines are probably the most generally used ligands in assymetric metallic catalysis for the synthesis of assorted helpful medication and prescription drugs. Have you ever ever questioned how you can get hold of them? A lot of the so-called chiral phosphines are C-stereogenic, which implies that the chirality is positioned at a web site apart from the P-center. Lots of them are at present commercially out there. Nevertheless, the true P-chiral phosphines, through which the chirality is discovered within the P-center itself, are probably the most troublesome to synthesize within the laboratory as a result of the formation of the C-P bond can’t be managed. Top-of-the-line identified strategies for C-P bond formation is a metal-catalyzed C-P cross-coupling reactions. Nevertheless, the management of chirality, i.e. the formation of a single chiral enantiomer, is difficult.
Researchers on the College of Groningen, led by 2016 Nobel Chemistry Prize winner Ben L. Feringa, have solved this downside of assymetric metallic catalyzed C-P bond formation by selecting an axially chiral 1,1’–bi–2–naphthol (BINOL)–based mostly phosphoramidite (also referred to as Feringas ligand) and aryl halides or triflates as beginning supplies, which was revealed in Nature Catalysis. The rationale for this success is the presence of BINOL, which controls the formation of a single isomer by a course of often called axial-central chirality switch. Though BINOLs are low cost (£38.00/10g in Fluorochem Uk), the researchers have additionally proven that it may be recycled with excessive yield and eantiopurity. “The largest problem in assymetric cross-coupling to kind C-P bonds is the aggressive ligand coordination between the exterior chiral phosphine ligand and the P-containing substrate/product. The important thing to our success is that the BINOL-containing phosphoramidites have the properties of an intrinsic chiral ligand and might function a substrate on the similar time,” provides one of many first authors, Anirban Mondal, a Ph.D. pupil in Feringa’s group.
Furthermore, the distinctive discovering of this work is the formation of an unexplored P-chiral phosphonium salt intermediate, genreted after arylation step, that may be visualized by X-ray crystallography. In keeping with the authors, this distinctive phosphonium salt, through which the chirality is positioned within the P-center, can open a brand new discipline as a chiral section switch catalyst or function a beginning materials for a lot of different P-chiral compounds.
“Once we first developed phosphoramidites as ligands in 1996, we have been intrigued by their wonderful stereocontrol in copper-catalyzed C-C bond formation, which led to a breakthrough in catalytic uneven conjugate addition. As phosphoramidites discovered use in trade, we imagined using them given their distinctive chiral properties,as beginning reagents for uneven transformations. Historically, an exterior chiral ligand is used for chiral induction in a C-P coupling response, however the aggressive coordination of preliminary and closing phosphorus compounds with the metallic catalysts, along with an exterior chiral ligand, reduces the enantioselectivity. As BINOL-containing phosphoramidites have the properties of an intrinsic chiral ligand and concurrently can function a substrate, we hypothesized that they’d improve stereoselectivity in C-P coupling processes with aryl compounds, and have been delighted when our information confirmed this,” provides Dr. Feringa in a analysis briefing revealed in Nature Catalysis.
The broad applicability and excessive flexibility of the phosphorus derivatives and artificial steps, along with an clever method to chirality switch by a budget and available BINOL methodology, open up a brand new route by which many difficult-to-access P-chirogenic compounds may be synthesized for a lot of functions, together with drug discovery, supplies chemistry, organoneucliotide chemistry, and particularly as ligands for metallic catalysis.
Mondal provides, “With this present methodology, we will now synthesize varied P-chiral phoshine ligands with each mirror picture varieties, opening up a brand new discipline of analysis. Finally, this may enable us to develop strategies through which P-chirality performs an vital position.”
Response kinetics drive chiral nanocrystal formation in tellurium atoms
Anirban Mondal et al, P-chirogenic phosphorus compounds by stereoselective Pd-catalysed arylation of phosphoramidites, Nature Catalysis (2021). DOI: 10.1038/s41929-021-00697-9
College of Groningen
Quotation:
New path to the synthesis of P-chiral compounds (2022, January 7)
retrieved 9 January 2022
from https://phys.org/information/2022-01-route-synthesis-p-chiral-compounds.html
This doc is topic to copyright. Aside from any truthful dealing for the aim of personal research or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for data functions solely.