Higher late than by no means, proper? Lastly, and after a number of delays, on Tuesday, Novavax (NVAX) introduced it had submitted to the FDA its request for Emergency Use Authorization (EUA) for Covid-19 vaccine NVX-CoV2373.
The setbacks the corporate had encountered on the trail towards the submitting principally revolved round manufacturing points. As such, B. Riley analyst Mayank Mamtani thinks a “key element” in driving approvals might be “confidence from authorities companies within the firm’s world manufacturing infrastructure to ship on the following demand.”
Till now, Novavax’s strategic accomplice, the Serum Institute of India (SII), has been accountable for the provision of doses. On the final rely, which incorporates the Indian authorities’s newest export allow of 250 million doses starting this month, the overall tally of doses allotted to be shipped from India has reached over 350 million. The majority of which has gone towards assembly the dedication to provide the COVAX facility with 1.1 billion doses of the vaccine. It at the moment stays unclear what the dose allocation plan is for upper-income nations.
Nonetheless, Mamtani notes that SII’s manufacturing capability, together with SK Biosciences’ “virtually comparable” progress on scale-up manufacturing, signifies Novavax is “gaining substantial provide to satisfy APA commitments to higher earnings nations, by way of CDMO preparations with these two strategic companions.” All this whereas the corporate ploughs forward with CMC information technology actions in 10+ North America and EU services.
Mamtani additionally thinks that because of the current vital “COVID illness burden” coupled with the “unequivocal real-world proof of diminished antibody-mediated vaccine efficacy,” the broadly held view that omicron has diminished the extent of urgency is misplaced. Accordingly, with “imminent” U.Okay. MHRA approval, adopted by the attainable US EUA, Novavax is well-positioned to supply the “solely different to mRNA vaccines in an effort to guarantee adequate immunization stage exists within the inhabitants previous to the emergence of subsequent COVID variant & ensuing wave of circumstances.”
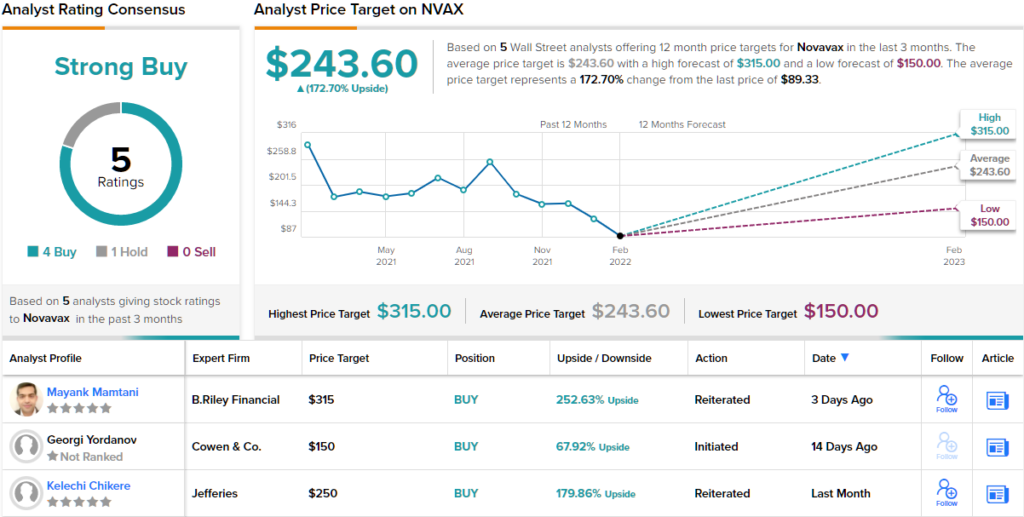
As such, Mamtani implores buyers make the most of an “enticing extra entry level,” to scoop up NVAX shares on a budget. The analyst’s Purchase score is backed by a $315 value goal, suggesting shares will rise by 231% within the 12 months forward. (To observe Mamtani’s observe file, click on right here)
Total, this vaccine maker will get a Robust Purchase score from the consensus of Wall Road’s analysts; the 5 current evaluations on the inventory embody 4 Buys and 1 Maintain. Novavax shares are promoting for $89.33, and the typical value goal of $243.60 suggests room for ~172% development within the 12 months forward. (See Novavax inventory forecast on TipRanks)

To seek out good concepts for shares buying and selling at enticing valuations, go to TipRanks’ Greatest Shares to Purchase, a newly launched device that unites all of TipRanks’ fairness insights.
Disclaimer: The opinions expressed on this article are solely these of the featured analyst. The content material is meant for use for informational functions solely. It is vitally essential to do your personal evaluation earlier than making any funding.