That’s the query {that a} latest JAMA research–Mulcahy et al. (2021)–purpose to reply. The authors use IQVIA MIDAS information to estimate gross sales value and quantity within the US. These can can be utilized to estimate drug wholesale acquisition value (WAC), however they ignore rebates. To account for rebates within the US they use SSR Well being info Then worldwide costs are recognized primarily based on the IQVIA MIDAS information throughout 6 markets (Australia, Canada, France, Germany, Japan, and the U.Ok). The authors then calculate US wholesale costs, US internet costs and what US costs can be if medicine have been reference priced to those 6 markets. Primarily based on this strategy, they discover that:
Worldwide reference pricing would have lowered 2020 US spending on research merchandise by 52.3% or $83.5 billion, from $159.9 billion at US internet costs to $76.3 billion…US net-to-international reductions ranged from 44.4% to 57.3% throughout therapeutic lessons
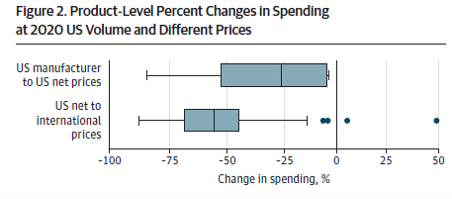
So if reference costs have been enacted as proposed in H.R. 3, may we count on a 50% lower in costs within the US? The reply is ‘no’. The rationale for that is that the Mulcahy et al. paper examines the impression of reference costs in a static fairly than a dynamic setting.
As famous in a latest Commonwealth Report, pharmaceutical corporations are more likely to alter their pricing methods if reference pricing have been adopted within the U.S. They could not settle for decrease costs in reference-priced nations or could delay entry in these markets.
The nations included within the reference basket may reply as effectively. Because the Commonwealth Fund report states:
…every of the six nations listed in H.R. 3 would have incentives to permit producers to exclude rebates and different value concessions from the (excessive) costs reported for incorporation into the AIM [average international market] value. The six nations would every profit by concealing the a lot decrease internet costs they might truly pay. Along with being of their nationwide curiosity, such guidelines additionally would profit producers and restrict drug financial savings within the U.S. by rising reported AIM and goal costs.
Additionally, launch costs could also be greater as effectively. Title II of H.R. 3 would impose rebates to offset costs that improve quicker than inflation. To insure the web current worth of a drug’s income of it’s lifetime stays related, it’s doubtless that pharmaceutical corporations would pursue excessive launch costs, if costs are extremely constrained over time.
Briefly, reference pricing is more likely to result in decrease costs within the U.S. (however not as a lot as anticipated by
Mulcahy and co-authors), greater costs and delays in entry for nations within the reference basket, and in addition the potential for fewer drug launches sooner or later if returns to life science R&D lower (see an August 2021 CBO report). There is no such thing as a free lunch.






