Among the many extra outstanding legacies of the covid-19 pandemic is how shortly federal regulators, the well being care trade, and customers moved to make at-home testing a dependable instrument for managing a public well being disaster.
However that fast-track focus is lacking from one other, much less publicized epidemic: an explosion in sexually transmitted illnesses that may trigger persistent ache and infertility amongst contaminated adults and disable or kill contaminated newborns. The disparity has amplified calls from researchers, public well being advocates, and well being care corporations urging the federal authorities to greenlight at-home testing kits that would vastly multiply the variety of People testing for STDs.
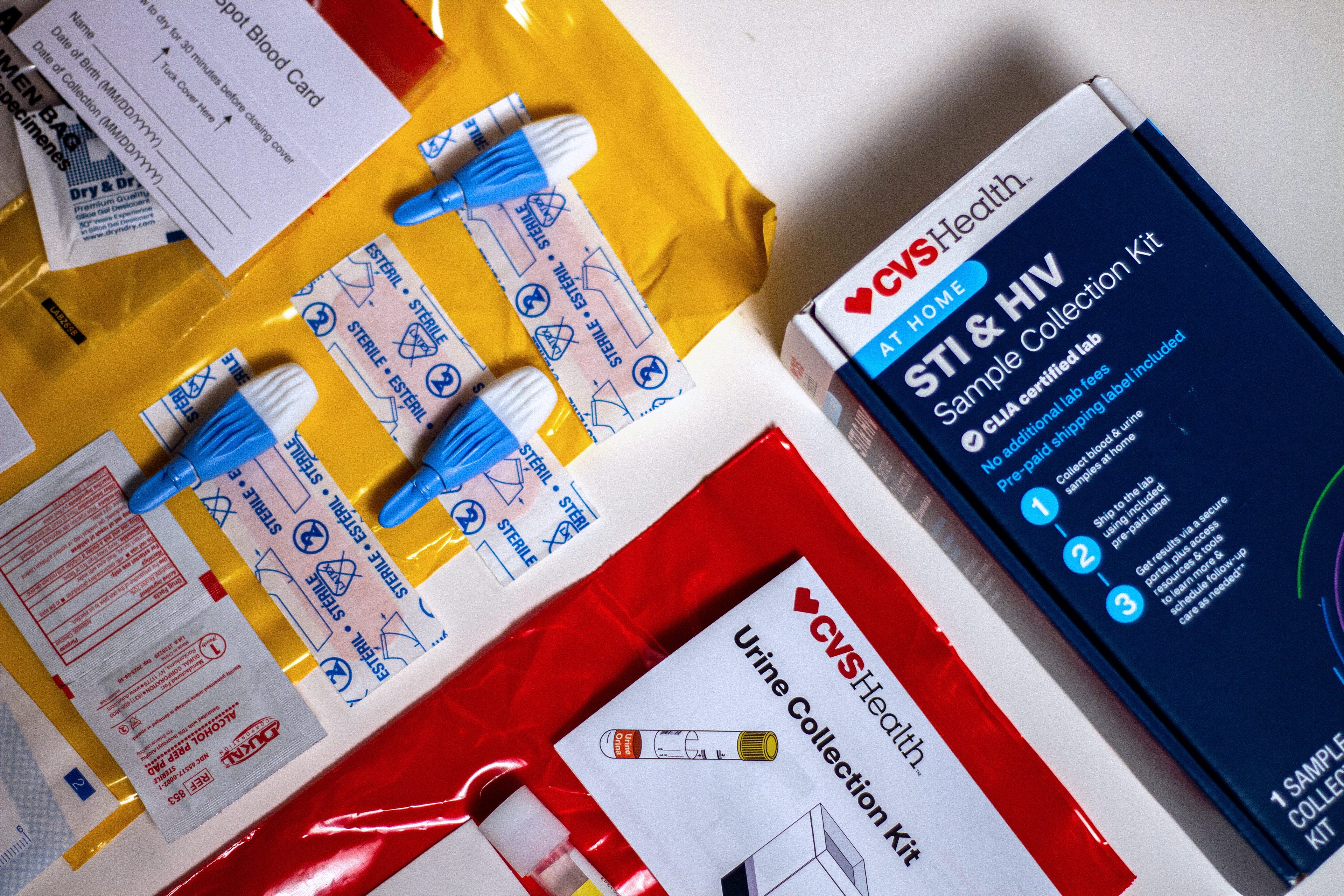
Internet buyers can already select from greater than a dozen self-testing kits, usually ranging in worth from $69 to $500, relying on the model and the number of infections they will detect.
However, apart from HIV assessments, the Meals and Drug Administration hasn’t authorized STD check kits to be used outdoors a medical setting. That leaves customers uncertain about their reliability whilst at-home use grows dramatically.
The STD epidemic is “uncontrolled,” mentioned Dr. Amesh Adalja, a senior scholar on the Johns Hopkins College Heart for Well being Safety. “We all know we’re lacking diagnoses. We all know that contact tracing is going on late or in no way. If we’re actually critical about tackling the STD disaster, we have now to get extra folks identified.”
Preliminary knowledge for 2021 confirmed almost 2.5 million reported circumstances of chlamydia, gonorrhea, and syphilis within the U.S., in line with the Facilities for Illness Management and Prevention. Reported circumstances of syphilis and gonorrhea have been climbing for a few decade. In its most up-to-date prevalence estimate, the company mentioned that on any given day, 1 in 5 People are contaminated with any of eight frequent STDs.
The push to make at-home testing for STDs as simple and commonplace as at-home covid and being pregnant testing is coming from a number of sectors. Public well being officers say their overextended staffers can’t deal with the staggering want for testing and surveillance. Diagnostic and pharmaceutical corporations see a enterprise alternative within the unmet demand.
The medical science underpinning STD testing will not be notably new or mysterious. Relying on the check, it could contain accumulating a urine pattern, pricking a finger for blood, or swabbing the mouth, genitals, or anus for discharge or cell samples. Medical facilities and neighborhood well being clinics have carried out such testing for many years.
The problem for regulators is whether or not sampling kits could be reliably tailored for in-home use. In contrast to speedy antigen assessments for covid, which produce leads to 15 to twenty minutes, the house STD kits available on the market require sufferers to gather their very own samples, after which bundle and mail them to a lab for evaluation.
Prior to now three years, because the pandemic prompted clinics that present low-cost care to drastically curtail in-person providers, a variety of public well being departments — amongst them state businesses in Alabama, Alaska, and Maryland — have began mailing free STD check kits to residents. Universities and nonprofits are additionally spearheading at-home testing efforts.
And dozens of business enterprises are leaping into or ramping up direct-to-consumer gross sales. Everly Well being, a digital well being firm that sells quite a lot of lab assessments on-line, reported gross sales for its suite of STD kits grew 120% within the first half of this yr in contrast with the primary half of 2021.
CVS Well being started promoting its personal bundled STD package in October, priced at $99.99. In contrast to most house kits, CVS’ model is stocked in shops.
Hologic, Abbott, and Molecular Testing Labs are among the many corporations urgently creating assessments. And Cue Well being, which sells covid assessments, is poised to launch a medical trial for a speedy house check for chlamydia and gonorrhea that will set a brand new bar, offering leads to about 20 minutes.
Alberto Gutierrez, who previously led the FDA workplace that oversees diagnostic assessments, mentioned company officers have been involved in regards to the reliability of house assessments for years. The FDA desires corporations to show that house assortment kits are as correct as these utilized in clinics, and that samples don’t degrade throughout transport.
“The company doesn’t consider these assessments are legally marketed at this level,” mentioned Gutierrez, a accomplice at NDA Companions, a consulting agency that advises corporations searching for to convey well being care merchandise to market.
“CVS shouldn’t be promoting that check,” he added.
In response to KHN questions, the FDA mentioned it considers house assortment kits, which may embody swabs, lancets, transport tubes, and chemical compounds to stabilize the samples, to be gadgets that require company evaluation. The FDA “usually doesn’t remark” on whether or not it plans to take motion on any particular case, the assertion mentioned.
CVS spokesperson Mary Gattuso mentioned the pharmacy chain is following the legislation. “We’re dedicated to making sure the merchandise we provide are protected, work as meant, adjust to laws, and fulfill clients,” Gattuso mentioned.
Everly Well being and different corporations described their kits as laboratory-developed assessments, akin to the diagnostics some hospitals create for in-house use. They usually contend their assessments could be legally marketed as a result of their labs have been licensed by a distinct company, the Facilities for Medicare & Medicaid Providers.
“The devices and assays utilized by the laboratories we use are similar to — and infrequently the identical as — these utilized by the labs a physician’s workplace makes use of,” mentioned Dr. Liz Kwo, chief medical officer at Everly Well being. “Our at-home pattern assortment strategies, like dried blood spots and saliva, have been extensively used for many years.”
Residence assortment kits enchantment to Uxmal Caldera, 27, of Miami Seashore, Florida, who prefers to check within the privateness of his house. Caldera, who doesn’t have a automobile, mentioned house testing saves him the time and expense of attending to a clinic.
Caldera has been testing himself for HIV and different STDs each three months for greater than a yr, a part of routine monitoring for folks taking PrEP, a routine of each day tablets to stop HIV an infection.
“Doing it by your self will not be exhausting in any respect,” mentioned Caldera, who’s uninsured however receives the assessments free by a neighborhood basis. “The directions are actually clear. I get the leads to perhaps 4 days. For certain, I’d advocate it to different folks.”
Dr. Leandro Mena, director of the CDC’s Division of STD Prevention, mentioned he want to see at-home STD testing develop into as routine as house being pregnant assessments. An estimated 16 million to twenty million assessments for gonorrhea and chlamydia are carried out within the U.S. annually, Mena mentioned. Widespread use of at-home STD testing, he mentioned, may double or triple that quantity.
He famous that medical doctors have years of expertise utilizing house assortment kits.
The Johns Hopkins Heart for Level-of-Care Applied sciences Analysis for Sexually Transmitted Illnesses has distributed roughly 23,000 at-home STD kits since 2004, mentioned Charlotte Gaydos, a principal investigator with the middle. The FDA usually permits such use if it’s a part of analysis overseen by medical professionals. The middle’s assessments are actually utilized by the Alaska well being division, in addition to Native American tribes in Arizona and Oklahoma.
Gaydos has revealed dozens of research establishing that house assortment kits for illnesses reminiscent of chlamydia and gonorrhea are correct and straightforward to make use of.
“There’s an enormous quantity of knowledge exhibiting that house testing works,” mentioned Gaydos.
However Gaydos famous that her research have been restricted to small pattern sizes. She mentioned she doesn’t have the tens of millions of {dollars} in funding it will take to run the form of complete trial the FDA usually requires for approval.
Jenny Mahn, director of medical and sexual well being on the Nationwide Coalition of STD Administrators, mentioned many public well being labs are reluctant to deal with house kits. “The general public well being labs gained’t contact it with out FDA’s blessing,” Mahn mentioned.
Public well being clinics typically present STD testing at little to no price, whereas medical health insurance usually covers in-person testing at a non-public observe. However most customers pay out-of-pocket for direct-to-consumer kits. Business pricing places them out of attain for many individuals, notably teenagers and younger adults, who account for almost half of STDs.
Adalja, at Johns Hopkins, mentioned the FDA has a historical past of transferring slowly on house testing. The company spent seven years evaluating the primary house HIV check it authorized, which hit the market in 2012.
“Residence testing is the best way of the long run,” mentioned Laura Lindberg, a professor of public well being at Rutgers College. “The pandemic opened the door to testing and remedy at house with out touring to a well being care supplier, and we aren’t going to have the ability to put the genie again within the bottle.”
KHN (Kaiser Well being Information) is a nationwide newsroom that produces in-depth journalism about well being points. Along with Coverage Evaluation and Polling, KHN is without doubt one of the three main working applications at KFF (Kaiser Household Basis). KFF is an endowed nonprofit group offering info on well being points to the nation.
USE OUR CONTENT
This story could be republished free of charge (particulars).